Controllable chemoselective hydrogenation of furfural by PdAg/C bimetallic catalysts under ambient operating conditions: an interesting Ag switch†
Abstract
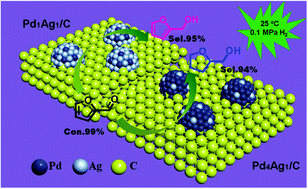
Hydrogenation of furfural to value-added chemical products is largely hindered by its multiple reaction pathways and complicated product distribution. Thus, to selectively achieve the desired products, catalysts with precise catalytic properties are highly required. Herein, a series of PdAg bimetallic nanoparticles (NPs) of similar size and tunable composition supported on activated carbon (Pd4Ag1/C, Pd2Ag1/C, Pd1Ag1/C and Pd2Ag3/C) were synthesized in a controlled manner and applied in the selective hydrogenation of furfural. Interestingly, an obvious composition-dependent catalytic performance was observed: upon incrementally increasing the proportion of Ag in PdAg NPs, the hydrogenation selectivity can transform from tetrahydrofurfuryl alcohol (sel. 94% for Pd4Ag1/C) to furfuryl alcohol (sel. 95% for Pd1Ag1/C) with nearly complete conversion (99%) of furfural. DFT calculations revealed that the adsorption free energy of in situ generated furfuryl alcohol on Pd(111) surface is inversely proportional correlated with the Ag content in PdAg bimetallic NPs, which accounts for the alteration of chemoselectivity. Importantly, the present study is the first demonstration of composition-induced selectivity reversal for the hydrogenation of furfural under ambient conditions (25 °C, 0.1 MPa H2).


 Please wait while we load your content...
Please wait while we load your content...