Aqueous electrosynthesis of silver indium selenide nanocrystals and their photothermal properties†
Abstract
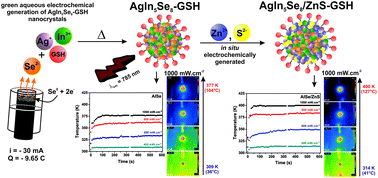
The eco-friendly aqueous electrosynthesis of AgIn5Se8 (AISe) nanocrystals (NCs), and their respective core/shell structure AgIn5Se8/ZnS NCs (AISe/ZnS), was carried out by using L-glutathione (GSH) as a stabilizer. Se0 direct reduction was carried out in a graphite powder macroelectrode of an electrochemical cavity cell, controlled by current (i = 30 mA) and electric charge (Q = 11.6 C). The generated Se2− ions react with In3+ and Ag+ ions in the intermediate compartment of the cell to give AISe NCs. A paired electrolysis (Zn0 anode/S0 in a graphite powder cathode) was carried out to prepare ZnS-GSH NCs, using the same electrochemical apparatus (i = 30 mA and Q = 9.6 C). The effects of the Ag+/In3+ ratio (0.5, 0.28 and 0.18) and ZnS shell on the optical properties of AISe NCs were investigated. The ternary AISe NCs were characterized by XRD and HRTEM, which revealed that AgIn5Se8 had a cubic phase structure (Ag+/In3+ = 0.18) and the ZnS shell had a zinc blende structure. The shell growth promoted a heterojunction formation with different crystalline phases, leading to a quantum yield decrease (QY: AISe = 18.6%; AISe/ZnS = 16.0%). The NCs were irradiated with a near-infrared laser at different power densities (λexc = 785 nm). AISe and AISe/ZnS samples showed photothermal response, reaching 381 K and 402 K, respectively.


 Please wait while we load your content...
Please wait while we load your content...