Smart and sustainable design of latent catalyst-containing benzoxazine-bio-resins and application studies†
Abstract
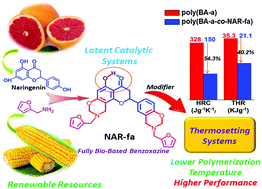
A straightforward synthetic approach to incorporate a hydrogen-bonding motif as part of a fully bio-based benzoxazine monomer (NAR-fa) is developed, leading to the first latent catalyst-containing thermosetting resin derived from natural renewable resources. The acronym is derived from the phenol (naringenin) and amine (furfurylamine) used in the synthesis. Interestingly, the newly developed benzoxazine resin exhibits a long shelf life in spite of possessing the lowest polymerization temperature reported hitherto for pure benzoxazines, 166 °C. The hydrogen-bonding motif is identified as an important design feature for studying the thermal behavior of the resin. All most common thermal and fire related properties, such as the glass transition temperature (Tg), temperature at which the weight loss is 5 and 10% (Td5 and Td10), char yield (Yc), limiting oxygen index (LOI), heat release capacity (HRC), and total heat released (THR), were far superior to those of typical polybenzoxazines. As a natural consequence of these great characteristics, NAR-fa was applied in small amounts (5 mol%) as an initiator and a property modifier of other petroleum-based and natural renewable resourced benzoxazine-based systems. The thermal properties of the resulting copolymeric thermosets were maintained or slightly enhanced, while those related to fire improved by about 38 and 51% for THR and HRC, respectively. These results highlight the utility of the latent catalyst-containing resin design derived from natural renewable resources in the preparation of high-performance resins and thermosets.


 Please wait while we load your content...
Please wait while we load your content...