Structure-performance correlations of cross-linked boronic acid polymers as adsorbents for recovery of fructose from glucose–fructose mixtures†
Abstract
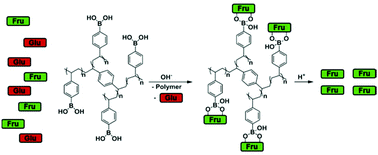
Recovery of bio-oxygenates from reaction mixtures is one of the major challenges for future bio-refineries. Isolation of fructose produced by isomerization of glucose presents a typical example at a very early stage of the value chain. We propose to recover fructose from a solution containing a mixture of glucose and fructose by adsorption on polymers bearing phenylboronate moieties. p-Vinylphenylboronic acid was polymerized with various cross-linkers, namely polar aliphatic, low-polarity aliphatic, and aromatic. The cross-linker content was in the range 5–40 mol%. The polymers exhibit high capacities for fructose, with a maximum loading of up to 1 molFru molB−1. Fructose loading depends significantly on the length and content of cross-linker, as well as pre-treatment of the polymer. In general, the maximum fructose capacity correlates with the swelling ability of the polymers, since phenylboronate moieties become available for adsorption upon swelling. In contrast, maximum glucose loadings are much lower, in the range 0.1–0.3 molGlu molB−1, and depend only slightly on the type of cross-linker. The structures of the glucose and fructose complexes and the kinetics of their uptake were studied by in situ MAS NMR. Efficient desorption of fructose was observed in acidic medium, and more importantly, using CO2. The structures of the polymers after repeated adsorption and desorption remain unchanged, as confirmed by solid-state NMR. Adsorption-assisted isomerization of glucose catalyzed by soluble carbonates was also studied. A 56% yield of fructose was achieved after 8 successive cycles of reaction and adsorption.


 Please wait while we load your content...
Please wait while we load your content...