A new strategy for CO2 utilization with waste plastics: conversion of hydrogen carbonate into formate using polyvinyl chloride in water†
Abstract
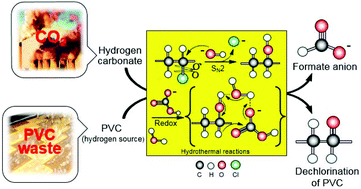
This work reports the first observation of the reduction of hydrogen carbonate to formate with polyvinyl chloride (PVC) as a hydrogen source in high-temperature water. Results show that NaHCO3 is successfully converted into formate with a 16% yield and nearly 100% selectivity based on hydrogen carbonate, simultaneously achieving the complete dechlorination of PVC at 300 °C. The mechanistic study suggests that CH–OH group formation from the substitution reaction of chlorine in PVC by OH− is an important starting step, and then the CH–OH group and HCO3− react to form formate via a redox reaction. Moreover, the selection of water as the reaction medium played a key role in the reduction of HCO3− to formate using PVC. The present study provides a new and green method for not only hydrogen carbonate/CO2 conversion but also PVC waste dechlorination in a single step.


 Please wait while we load your content...
Please wait while we load your content...