The pattern of peptides released from dairy and egg proteins is highly dependent on the simulated digestion scenario
Abstract
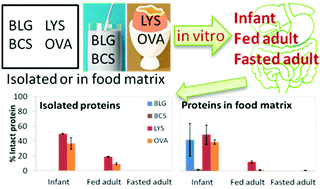
Evaluating the gastrointestinal (GI) fate of proteins is part of the assessment to determine whether proteins are safe to consume. In vitro digestion tests are often used for screening purposes in the evaluation of potential allergenicity. However, the current pepsin resistant test used by the European Food Safety Authority, only corresponds to fasted gastric conditions representative of a late phase adult stomach. In addition, these tests are performed on isolated proteins and the effect of the food matrix and processing are not systematically considered. The aim of this research is to compare three different static in vitro GI scenarios that are physiologically relevant. Namely, an infant, early phase (fed state) adult and late phase (fasted state) adult model. These protocols are applied to well-characterised isolated dairy (β-lactoglobulin and β-casein) and egg (lysozyme and ovalbumin) proteins and the impact of food matrix/processing on their proteolysis is also investigated. A combination of SDS-PAGE, LC-MS/MS and spectrophotometric assay was used for the evaluation of the proteolysis. Results highlight differences across the three GI scenarios whether on isolated proteins or within food matrices. The infant model led to incomplete digestion, leaving intact egg proteins, either isolated or in the food matrix, and intact β-lactoglobulin in the milk. In addition, peptides greater than 9 amino acids were found throughout the intestinal phase for all proteins studied, regardless of the scenario. This reinforces the difficulty of linking protein digestibility to potential allergenicity because many other factors are involved that need further investigation.
- This article is part of the themed collection: Food & Function Recent Open Access Articles


 Please wait while we load your content...
Please wait while we load your content...