Theoretical and experimental investigation of the antioxidative activity of monascin
Abstract
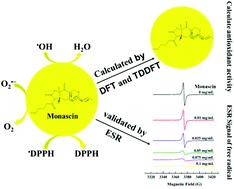
Monascin, a specific type of monascus pigments, exhibits many bioactivities. In this study, the antioxidative activity of monascin was investigated by theoretical and experimental methods. First, the antioxidant potential of six monascus pigments was predicted by density functional theory (DFT) and time-dependent density functional theory (TDDFT) at the B3LYP/6-31+G (d, p) level, and monascus yellow pigments were predicted to have strong antioxidant capacity, as they can transfer hydrogen to free radicals and accept electrons from radicals. Then, the free radical-scavenging capacity of monascin for 2,2-diphenyl-1-picrylhydrazyl (DPPH), superoxide, and hydroxyl radicals was experimentally validated by electron spin resonance (ESR) measurement. Monascin exhibited a quenching effect on DPPH, superoxide, and hydroxyl radicals in a dose-dependent manner. Specifically, the scavenging activity of monascin for DPPH, superoxide, and hydroxyl radicals was 97.5%, 59.5%, and 68.6%, respectively, when 0.1 mg mL−1 monascin was present. Our study provides theoretical evidence for the strong antioxidative activity of monascin and offers a simple and reliable strategy to determine the antioxidative activity.
- This article is part of the themed collection: Food & Function Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...