Acetylcholinesterase inhibitory activity and neuroprotection in vitro, molecular docking, and improved learning and memory functions of demethylcurcumin in scopolamine-induced amnesia ICR mice
Abstract
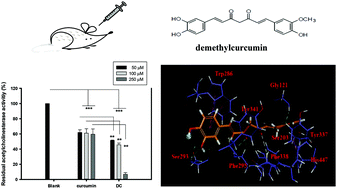
In this study, demethylcurcumin (DC), a minor constituent in curcuminoids, showed better anti-acetylcholinesterase (anti-AChE) activities, anti-amyloid β peptide aggregation, neuroprotective activities in 6-hydroxydopamine-treated SH-SY5Y cell models, and anti-nitric oxide production in lipopolysaccharide-treated RAW 264.7 macrophages than those of curcumin. Based on molecular docking analyses with AChE, the meta-hydroxyl group in DC, nonexistent in curcumin, showed the formation of hydrogen bonds with Ser293 and Tyr341 in the binding sites of AChE. For animal experiments, scopolamine-induced amnesia ICR mice were used to analyze the learning and memory functions of DC in comparison with the positive control donepezil. Mice fed with DC (50 mg kg−1) or donepezil (5 mg kg−1) showed improvement and a significant difference compared to those in the control group (P < 0.05, 0.01, or 0.001) in a passive avoidance test and in a water maze probe test. The brain extracts of the mice in the DC or donepezil group showed reduced AChE activities and higher ORAC activities and also showed a significant difference compared to those in the control group (P < 0.05, 0.01, or 0.001). DC might be beneficial for developing functional foods or as a lead compound for the treatment of degenerative disorders.


 Please wait while we load your content...
Please wait while we load your content...