Structural analysis and allergenicity assessment of an enzymatically cross-linked bovine α-lactalbumin polymer
Abstract
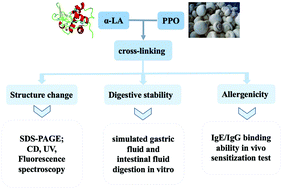
Enzymatic cross-linking is frequently used in bio-processing of dairy products since it could change the physiochemical and functional characterization. In our study, bovine α-lactalbumin was cross-linked by polyphenol oxidase from Agaricus bisporus and the changes in the structure, digestibility and allergenicity of α-lactalbumin were explored after cross-linking, and the structural alterations of the polymers were analyzed by circular dichroism spectroscopy, ultraviolet absorption spectroscopy and fluorescence spectroscopy. The digestibility of cross-linked α-lactalbumin was evaluated by simulated digestion in vitro. After that, the allergenicity of α-lactalbumin polymers was evaluated by detection of the specific IgE binding ability using an animal model. The results showed that the secondary and tertiary structures of various α-lactalbumin polymers exhibited a significant variation compared with those of untreated α-lactalbumin, and the cross-linked α-lactalbumin was relatively less susceptible to digestion. Moreover, the allergenicity of cross-linked polymers decreased significantly. These results suggested that there was a direct correlation between a loss of an α-helix and IgE binding to α-lactalbumin, which indicated that enzymatic cross-linking might be an efficient approach to reduce the allergenicity of bovine α-lactalbumin.


 Please wait while we load your content...
Please wait while we load your content...