Iron encapsulated microstructured gel beads using an emulsification–gelation technique for an alginate-caseinate matrix
Abstract
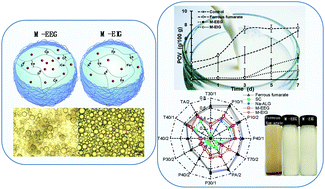
Iron-deficiency anemia is an important health problem in global public issues, and development of iron fortifiers in diets is essential for the decrease of iron deficiency. However, there are problems for iron fortification in food because the common bioavailable iron compounds would contribute to iron-promoted lipid oxidation and unpleasant iron odor, presenting an adverse food quality. Ferrous fumarate loaded microstructured gel beads were prepared by an emulsification–gelation method using an alginate-caseinate matrix, and the gel network was formed by crosslinking of Ca2+ or Fe2+. Internal gelated beads showed relatively symmetrical and homogeneous spheres with no adhesion due to the simultaneous release of Fe2+ to initiate gelation in situ. External gelated beads displayed an irregular and adhesive structure, probably because the random contact between Na-ALG and Ca2+ occurred on the droplet surface, and the immediately gelated hardening layer provided a delay for further Ca2+ diffusion. The gel beads exhibited a lag phase in the promotion of lipid oxidation of the emulsion and restrained the iron odor release from ferrous fumarate. Ferrous ion release from microstructured gel beads in the simulated gastric juice was obviously delayed before a more progressive high release in the simulated intestinal juice, beneficial for iron absorption in the duodenum. The iron encapsulated microstructured gel beads might be developed as a promising safe iron fortifier by relieving lipid oxidation and iron odor.


 Please wait while we load your content...
Please wait while we load your content...