β-Sitosterol regulated microRNAs in endothelial cells against an oxidized low-density lipoprotein
Abstract
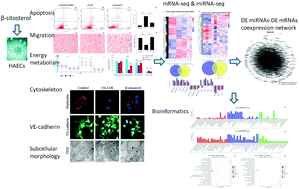
β-Sitosterol is a natural compound widely found in many vegetable oils, nuts, and plant medicines; it lowers the cholesterol levels, enhances the production of plasminogen activators, and exhibits anticancer and antiatherogenic effects. However, the direct endothelial protection of β-sitosterol against an oxidized low-density lipoprotein (ox-LDL) is not well understood. In the present study, β-sitosterol significantly inhibited cell apoptosis (P < 0.01), increased cell migration (P < 0.01), improved energy metabolism (P < 0.05) and improved morphology after ox-LDL (50 μg ml−1) exposure following β-sitosterol (2 μg mL−1) treatment in human aortic endothelial cells (HAECs ). A total of 691 differentially expressed (DE) mRNAs were identified (579 were upregulated and 112 were downregulated, fold change ≥2.0, P < 0.05) after 24 h of β-sitosterol administration in transcriptome sequencing (β-sitosterol vs. ox-LDL), which suggested that β-sitosterol reversed 62.32% change in mRNAs induced by ox-LDL. DE mRNAs are enriched mainly in focal adhesion, ribosomes, eukaryotic translation elongation, etc. Considering that one of the enrichment is 3′-UTR-mediated translational regulation, we explored DE microRNA (miRNA). The miRNA-seq data proposed 87 up-regulated and 58 down-regulated miRNAs (fold change ≥2.0, P < 0.05) in miRNA-seq (β-sitosterol vs. ox-LDL), suggesting that β-sitosterol reversed 76.67% change in miRNAs induced by ox-LDL. The DE miRNA-DE mRNA coexpression network focused on ribosomes, cell cycle, oxidative phosphorylation, PI3K-Akt signaling pathway, TNF signaling pathway, ErbB signaling pathway, and mTOR signaling pathway. Consequently, miRNAs might be the targets of β-sitosterol and play vital roles in transcriptional regulation in endothelial protective and antiatherogenic effects against ox-LDL.


 Please wait while we load your content...
Please wait while we load your content...