Order-disorder transitions of cytoplasmic N-termini in the mechanisms of P-type ATPases†
Abstract
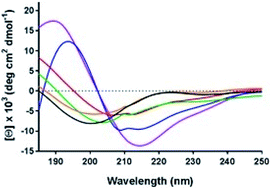
Membrane protein structure and function are modulated via interactions with their lipid environment. This is particularly true for integral membrane pumps, the P-type ATPases. These ATPases play vital roles in cell physiology, where they are associated with the transport of cations and lipids, thereby generating and maintaining crucial (electro-)chemical potential gradients across the membrane. Several pumps (Na+, K+-ATPase, H+, K+-ATPase and the plasma membrane Ca2+-ATPase) which are located in the asymmetric animal plasma membrane have been found to possess polybasic (lysine-rich) domains on their cytoplasmic surfaces, which are thought to act as phosphatidylserine (PS) binding domains. In contrast, the sarcoplasmic reticulum Ca2+-ATPase, located within an intracellular organelle membrane, does not possess such a domain. Here we focus on the lysine-rich N-termini of the plasma-membrane-bound Na+, K+- and H+, K+-ATPases. Synthetic peptides corresponding to the N-termini of these proteins were found, via quartz crystal microbalance and circular dichroism measurements, to interact via an electrostatic interaction with PS-containing membranes, thereby undergoing an increase in helical or other secondary structure content. As well as influencing ion pumping activity, it is proposed that this interaction could provide a mechanism for sensing the lipid asymmetry of the plasma membrane, which changes drastically when a cell undergoes apoptosis, i.e. programmed cell death. Thus, polybasic regions of plasma membrane-bound ion pumps could potentially perform the function of a “death sensor”, signalling to a cell to reduce pumping activity and save energy.
- This article is part of the themed collection: Peptide-membrane interactions


 Please wait while we load your content...
Please wait while we load your content...