Exploring cooperative porosity in organic cage crystals using in situ diffraction and molecular simulations†
Abstract
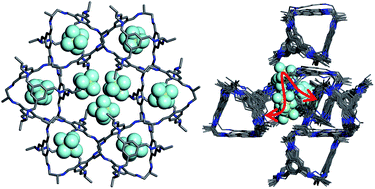
A porous organic cage crystal, α-CC2, shows unexpected adsorption of sulphur hexafluoride (SF6) in its cage cavities: analysis of the static crystal structure indicates that SF6 is occluded, as even the smallest diatomic gas, H2, is larger than the window of the cage pore. Herein, we use in situ powder X-ray diffraction (PXRD) experiments to provide unequivocal evidence for the presence of SF6 inside the ‘occluded’ cage voids, pointing to a mechanism of dynamic flexibility of the system. By combining PXRD results with molecular dynamics simulations, we build a molecular level picture of the cooperative porosity in α-CC2 that facilitates the passage of SF6 into the cage voids.
- This article is part of the themed collection: Cooperative phenomena in framework materials


 Please wait while we load your content...
Please wait while we load your content...