Photoelectrochemical alcohol oxidation by mixed-linker metal–organic frameworks†
Abstract
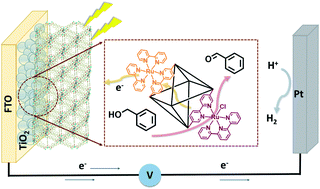
Metal–organic frameworks (MOFs) provide a suitable platform for stable and efficient heterogeneous photoelectrochemical oxidation catalysis due to their highly ordered structure, large surface area, and synthetic tunability. Herein, a mixed-linker MOF comprising of a photosensitizer [Ru(dcbpy)(bpy)2]2+ (bpy = 2,2′-bipyridine, dcbpy = 5,5′-dicarboxy-2,2′-bipyridine) and catalyst [Ru(tpy)(dcbpy)Cl]+ (tpy = 2,2′:6′,2′′-terpyridine) that were incorporated into the UiO-67 framework and grown as thin films on a TiO2-coated, fluorine-doped tin oxide (FTO) electrode (RuB-RuTB-UiO-67/TiO2/FTO). When used as an electrode for the photoelectrochemical oxidation of benzyl alcohol, the mixed-linker MOF film showed a faradaic efficiency of 34%, corresponding to a 3-fold increase in efficiency relative to the RuB-UiO-67/TiO2/FTO control. This increase in catalytic efficiency is ascribed to the activation of RuTB moieties via oxidation by photogenerated RuIIIB. Transient absorption spectroscopy revealed the delayed appearance of RuIIITB* or RuIIITB formation, occurring with a lifetime of 21 ns, due to energy and/or electron transfer. The recovery kinetics of the charge separated state was increased (283 μs) in comparison to single-component control experiments (105 μs for RuB-UiO-67/TiO2/FTO and 7 μs for RuTB-UiO-67/TiO2/FTO) indicating a cooperative effect that could be exploited in chromophore/catalyst MOF motifs.
- This article is part of the themed collection: Cooperative phenomena in framework materials


 Please wait while we load your content...
Please wait while we load your content...
