A combined periodic DFT and QM/MM approach to understand the radical mechanism of the catalytic production of methanol from glycerol†
Abstract
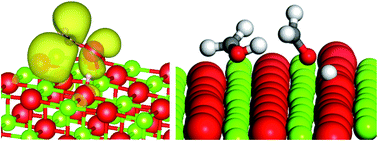
The production of methanol from glycerol over a basic oxide, such as MgO, using high reaction temperatures (320 °C) is a promising new approach to improving atom efficiency in the production of biofuels. The mechanism of this reaction involves the homolytic cleavage of the C3 feedstock, or its dehydration product hydroxyacetone, to produce a hydroxymethyl radical species which can then abstract an H atom from other species. Obtaining a detailed reaction mechanism for this type of chemistry is difficult due to the large number of products present when the system is operated at high conversions. In this contribution we show how DFT based modelling studies can provide new insights into likely reaction pathways, in particular the source of H atoms for the final step of converting hydroxymethyl radicals to methanol. We show that water is unlikely to be important in this stage of the process, C–H bonds of C2 and C3 species can give an energetically favourable pathway and that the disproportionation of hydroxymethyl radicals to methanol and formaldehyde produces a very favourable route. Experimental analysis of reaction products confirms the presence of formaldehyde. The calculations presented in this work also provide new insight into the role of the catalyst surface in the reaction showing that the base sites of the MgO(100) are able to deprotonate hydroxymethyl radicals but not methanol itself. In carrying out the calculations we also show how periodic DFT and QM/MM approaches can be used together to obtain a rounded picture of molecular adsorption to surfaces and homolytic bond cleavage which are both central to the reactions studied.
- This article is part of the themed collection: Reaction mechanisms in catalysis


 Please wait while we load your content...
Please wait while we load your content...