Computational design of graphitic carbon nitride photocatalysts for water splitting†
Abstract
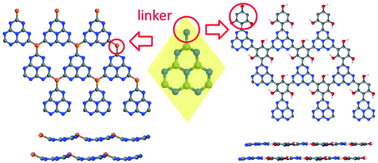
A series of structures based on graphitic carbon nitride (g-C3N4), a layered material composed of linked carbon–nitrogen heterocycles arranged in a plane, were investigated by density functional theory calculations. g-C3N4 is a semiconductor that absorbs UV light and visible light at the blue end of the visible spectrum, and is widely studied as a photocatalyst for water splitting; however, its photocatalytic efficiency is limited by its poor light-harvesting ability and low charge mobilities. Modifications to the g-C3N4 structure could greatly improve its optical and electronic properties and its photocatalytic efficiency. In this work, the g-C3N4 structure was modified by replacing the nitrogen linker with heteroatoms (phosphorus, boron) or aromatic groups (benzene, s-triazine and substituted benzenes). Two-dimensional (2D) sheets and three-dimensional (3D) multilayer structures with different stacking types were modelled. Several new structures were predicted to have electronic properties superior to g-C3N4 for use as water splitting photocatalysts. In particular, introduction of phosphorus, benzene and s-triazine groups led to band gaps smaller than in the standard g-C3N4 (down to 2.4 eV, corresponding to green light). Doping with boron in the linker positions dramatically reduced the band gap (to 1.6 eV, corresponding to red light); the doped material has the valence band position suitable for water oxidation. Our computational study shows that chemical modification of g-C3N4 is a powerful method to tune this material’s electronic properties and improve its photocatalytic activity.
- This article is part of the themed collection: Chemistry of 2-dimensional materials: beyond graphene


 Please wait while we load your content...
Please wait while we load your content...