Supported FexNiy catalysts for the co-activation of CO2 and small alkanes†
Abstract
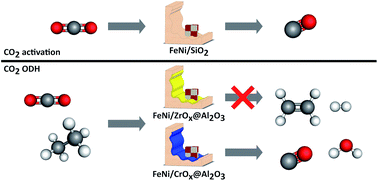
The effect of both the Fe : Ni ratio (5 to 1 : 1) and the relative Lewis acidity of a metal oxide support on catalytic activity, selectivity and stability was investigated in the CO2 mediated oxidative dehydrogenation of ethane (CO2-ODH). To avoid effects of varying pore sizes, shapes and volumes of the supports, chromia and zirconia overlayers were coated onto a common γ-Al2O3 carrier (CrOx@Al2O3 and ZrOx@Al2O3). Separately, oxidic FexNiy alloy precursor nanoparticles were prepared using a nonaqueous surfactant-free method and deposited by sonication onto the carrier. In comparison to previous studies in the field, this synthesis technique yields closely associated iron and nickel increasing the chances for alloy formation. During reduction, a mixture of a bcc and a fcc alloy phase was formed, with the content of bcc increasing with increasing iron content as predicted by the bulk phase diagram. Upon exposure to carbon dioxide at elevated temperatures, the bcc metallic phase is selectively oxidised to an inverse spinel structure via the dissociation of CO2. When exposed to CO2-ODH conditions, the bare ZrOx@Al2O3 support shows no activity. The presence of FeNi phases increases the conversion of ethane and CO2 marginally (<2%) but forms ethylene at high selectivity (SC2H4 > 80%). The CrOx@Al2O3 support shows some initial activity (XC2H6 < 5%) at very high ethylene selectivity (SC2H4 > 90%) but deactivates with time on stream. Comparison of the ethane and carbon dioxide conversions suggests that direct dehydrogenation rather than the oxidative pathway is taking place. When FeNi particles with the highest Fe content are added, the ethane conversion behavior hardly changes, but the CO2 conversion is increased now supporting the stoichiometric CO2-ODH reaction (SC2H4 > 95%). It is therefore evident that a tandem catalyst system between a reducible oxide carrier and the FeNi species is required. Increasing the Ni content results in an increase in activity and stability while changing the dominant reaction pathway to a combination of dry reforming, CO2-ODH and possibly the reverse Boudouard reaction, with the latter countering catalyst deactivation through carbon deposition.
- This article is part of the themed collection: Reaction mechanisms in catalysis


 Please wait while we load your content...
Please wait while we load your content...