Graphene-supported 2D cobalt oxides for catalytic applications
Abstract
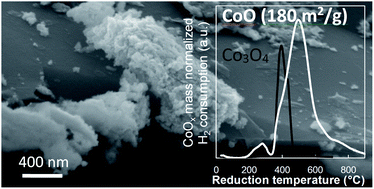
2D materials are attracting increasing attention in many strategic applications. In particular, ultra-thin non-layered oxides have been shown to outperform their 3D counter-parts in several health and energy applications, such as the removal of toxic carbon monoxide by low temperature oxidation and the development of high performance supercapacitors. The general reason for that is the increased surface-to-volume ratio, which maximizes exposure of active species and enhances exchange between the (limited) bulk and the surface. The challenge is to synthesize such 2D configurations of 3D oxides, which generally requires quite harsh multi-step, multi-reagent chemical processes. Here we show that natural graphite can be used as a templating matrix to grow non-stoichiometric 2D transition metal oxides. We focus on highly porous, highly reduced cobalt oxides grown from cobalt nitrate and sodium borohydride under sonication. Extensive characterization, including nitrogen physisorption, thermogravimetric analysis (TGA), scanning and transmission electron microscopy (SEM/TEM), X-ray diffraction (XRD), temperature programmed oxidation and reduction (TPO/TPR), Fourier transformed infrared (FTIR) and Raman spectroscopies, highlights the specific features of the 2D morphologies (nanosheets and nanofilms) obtained. For comparison, 3D morphologies of Co3O4 spinel nanocrystallites are grown from stacked 2D cobalt phthalocyanine–graphene precursors upon controlled thermal oxidation. Finally, low temperature CO oxidation catalysis evidences the superior performance of the graphene-supported CoO-like cobalt oxide 2D nanosheets.
- This article is part of the themed collection: Chemistry of 2-dimensional materials: beyond graphene


 Please wait while we load your content...
Please wait while we load your content...