Modulating donor–acceptor transition energies in phosphorus–boron co-doped silicon nanocrystals via X- and L-type ligands†
Abstract
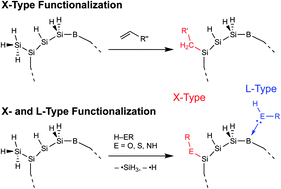
In this work, we explore the effect of ligand binding groups on the visible and NIR photoluminescent properties within phosphorus–boron co-doped silicon nanocrystals (PB:Si NCs) by exploiting both the X-type (covalent) and L-type (Lewis donor molecule) bonding interactions. We find that the cooperative nature of both X- and L-type bonding from alkoxide/alcohol, alkylamide/alkylamine, and alkylthiolate/alkylthiol on PB:Si NCs results in photoluminescence (PL) energy blue shifts from the as-synthesized, hydride-terminated NCs (PB:Si–H) in excess of 0.4 eV, depending on the surface termination. These PL blue shifts appear greatest in the most strongly confined samples with diameters <4 nm where the surface-to-volume ratio is high and, therefore, the ligand effects are most pronounced. A correlation between the donor group strength (either X-type or L-type) and the degree of D–A state modulation is found, and the proportion of the PL blue shift from the X- and L-type interactions is quantified. Raman spectroscopy is used to provide additional evidence of the strength of the L-type donor groups. Additionally, we probe how the nature of the ligand chemistry affects the radiative lifetime and PL efficiency and find that the ligands do not significantly change the D–A emission dynamics, and all samples retain the long 50–130 μs lifetimes characteristic of these transitions. Finally, we describe three mechanisms that operate to affect the D–A recombination energies: (1) X-type ligands that modulate the PB:Si–X NC wavefunction; (2) L-type ligands that perturb the donor and acceptor states via a molecular orbital theory picture; and (3) X- and L-type ligands that cause a dielectric increase around the PB:Si NC core, which provides Coulomb screening and modulates the donor and acceptor states even further.
- This article is part of the themed collection: Luminescent silicon nanostructures


 Please wait while we load your content...
Please wait while we load your content...