Linear solvation energy relationship development for adsorption of synthetic organic compounds by carbon nanomaterials: an overview of the last decade
Abstract
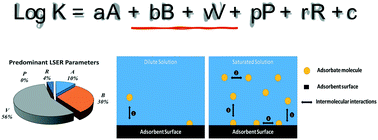
The production of carbon nanomaterials has been increasing due to their outstanding properties and innovation potential in various industrial applications. Hence, their discharge into the aquatic environment from point and non-point sources is inevitable. Therefore, the natural and built aquatic environments that contain thousands of synthetic organic contaminants, would facilitate adsorptive interaction between synthetic organic molecules and carbon nanomaterial surfaces. Today's literature rapidly developed to contain a great number of adsorption data for synthetic organic compounds onto carbon nanomaterials. The availability of adsorption data enables generation of structure–activity relationships by utilizing adsorbate and adsorbent characteristics in simple/controlled aquatic solutions. These models not only create practical predictive tools but also provide mechanistic insights about intermolecular interactions between organic molecules and carbon nanomaterials. This review analyzes 86 linear solvation energy relationship models published in the last decade. Our work summarizes and compares key independent variables associated with adsorbate properties (e.g., molecular size, H-bonding ability, polarizability) and identifies critical factors for selecting dependent variables (e.g., adsorbent characteristics, saturation concentration). The study also discusses intermolecular interactions unraveled by the modeling efforts at different adsorbate concentrations. This work aims to provide an analysis of literature to identify key gaps in today's model training efforts and list guiding principles for ongoing and future linear solvation energy relationship studies.


 Please wait while we load your content...
Please wait while we load your content...