Comparison of the impact of ozone, chlorine dioxide, ferrate and permanganate pre-oxidation on organic disinfection byproduct formation during post-chlorination†
Abstract
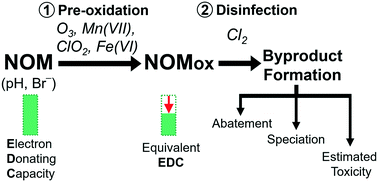
Pre-oxidation is commonly used to mitigate the formation of byproducts during post-disinfection. A comparative study of the impact of four pre-oxidants, ozone (O3), chlorine dioxide (ClO2), permanganate (Mn(VII)) and ferrate (Fe(VI)), on the formation of trihalomethanes (THMs), haloacetonitriles (HANs) and adsorbable organic halogens (AOX) in chlorinated synthetic and real waters was conducted. The pre-oxidant doses were based on their impact on natural organic matter reactivity measured by the electron donating capacity before chlorination. The influence of pH (6.5–8.1) and bromide (0–500 μg L−1) was evaluated in terms of disinfection byproduct (DBP) formation and theoretical toxicity assessment (based on THM and HAN formation). All oxidants were efficient in mitigating chlorinated DBPs, except Mn(VII) which had little impact on THM formation. O3 was generally more efficient than the other oxidants in mitigating AOX and THM formation, all pre-oxidants readily reduced the formation of HANs (>45% reduction at high dose). pH depression improved AOX mitigation by O3 and Fe(VI) but diminished Mn(VII) efficiency for all DBPs. Pre-oxidation was less efficient in mitigating brominated DBPs and generally enhanced the bromine substitution factor. Although HANs were formed at low concentrations compared to THMs, they dominated the calculated toxicity, particularly the brominated HANs. The increased dibromoacetonitrile formation after pre-oxidation was a major factor counteracting the benefits of the overall DBP mitigation. In the presence of bromide, the pre-oxidant dose should be optimized to decrease the reactivity of the matrix while controlling the toxicity induced by formation of brominated DBPs, notably brominated HANs.
- This article is part of the themed collection: Drinking water oxidation and disinfection processes


 Please wait while we load your content...
Please wait while we load your content...