Characterization of a commercially-available, low-pressure UV lamp as a disinfection system for decontamination of common nosocomial pathogens on N95 filtering facepiece respirator (FFR) material†
Abstract
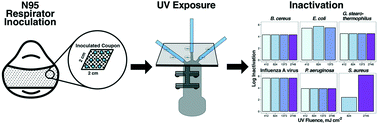
A commercially-available UV surface disinfection system used for hospital room disinfection was characterized for use as a UV-inactivation system for N95 filtering facepiece respirator (FFR) respirators. The output of light was initially characterized and assessed for its ability to penetrate through the individual N95 respirator layers. Following characterization, disinfection performance was tested using a range of model pathogens. In a series of experiments, coupons of respirator material were inoculated with Escherichia coli, Staphylococcus aureus, Geobacillus stearothermophilus spores, Bacillus cereus, Pseudomonas aeruginosa, and influenza A virus. Inoculated coupons were then treated with UV light from a low-pressure UV disinfection system during specific treatment times. Microbial enumeration was performed pre- and post-UV treatment. It was found that respirator coupons inoculated with E. coli, influenza A, P. aeruginosa, B. cereus and G. stearothermophilus resulted in below detection counts when exposed to UV fluences between 410 to 2700 mJ cm−2. In addition, UV light was unable to sufficiently penetrate all layers of respirator material, highlighting the importance of flipping respirators when exposing them to UV light. This work suggests that with further validation, UV light will be a promising touchless treatment option for frontline professionals who need to repurpose N95 respirators for subsequent use.
- This article is part of the themed collections: Coronavirus articles - free to access collection and Environmental Science – coronavirus research


 Please wait while we load your content...
Please wait while we load your content...