Purification and removal of the low molecular weight fraction of polyDADMAC reduces N-nitrosodimethylamine formation during water treatment†
Abstract
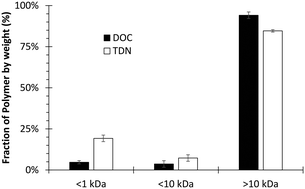
Cationic polymers are critical coagulant aids at drinking water plants, but the same polymers are simultaneously N-nitrosodimethylamine (NDMA) precursors upon chloramine exposure. We hypothesized the low-molecular fraction, which represents a small mass residual in polyDADMAC cationic polymer, reacts with chloramines, may not be well removed during coagulation, and is thus responsible for forming parts-per-trillion (ppt) concentrations of NDMA in finished water. To identify strategies capable of reducing post-coagulation residual polymer associated with NDMA formation, this study fractionated polyDADMAC by molecular weight, characterized reactivity of each fraction, and quantified precursor contributions to NDMA formation potential during bench-scale test and NDMA concentration due to formation during treatment during pilot tests. Diaultrafiltration of the cationic polymer produced a low molecular weight fraction (<10 kDa) and a purified polyDADMAC (>10 kDa). Native organic matter in bench- and pilot- tests contributed 19–38% of NDMA formed throughout treatment, while polymers were responsible for 62–81% of NDMA formed. The <10 kDa fraction of polyDADMAC was more reactive (450–540 ng NDMA per mg DOC) and formed >10× NDMA than non-purified polyDADMAC in jar tests. Purified polyDADMAC, with <10 kDa fraction removed, formed 54% less NDMA during pilot tests (and 63% less during jar tests) than non-purified polyDADMAC. There was no adverse effect on coagulation or subsequent filter performance by using purified polyDADMAC, but significantly (p < 0.05) less NDMA formed in filtered water when using diaultrafiltered cationic polymer. Thus, removing the low molecular weight impurities (<10 kDa) by polymer suppliers would lead to an equally-effective coagulant-aid that substantially lowers NDMA formation during drinking water treatment.
- This article is part of the themed collection: Drinking water oxidation and disinfection processes


 Please wait while we load your content...
Please wait while we load your content...