Removal of contaminants from canal water using microwave synthesized zero valent iron nanoparticles†
Abstract
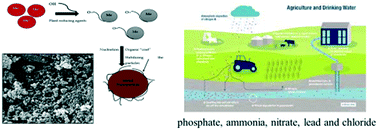
Nanoscale zero valent iron nanoparticles (nZVI-NPs) have been used for the remediation of a wide variety of environmental contaminants. Here, nZVI-NPs were synthesized using a green method involving leaf extract of Mentha piperita as the reducing agent and microwave treatment in place of conventional heating. The resulting NP composition, morphology, surface charge and size were studied using UV-visible spectrophotometry (UV-vis), scanning electron microscopy coupled with energy dispersive X-ray spectroscopy (SEM-EDX) and dynamic light scattering (DLS), respectively. The plasmon resonance spectrum of the synthesized nZVI-NPs had a peak at 292 nm. The synthesized nZVI-NPs were spherical in shape, with sizes between 5 and 10 nm, and negatively charged with zeta potential of −27.9 ± 2.19 mV. The nZVI-NPs with a high reactive surface area were successfully utilized to remove phosphate, lead, ammonia, nitrate and chloride from water samples taken from the Worcester and Birmingham canal at the University of Birmingham, United Kingdom. A successful rapid removal of dissolved agricultural contaminants (phosphate, ammonia, nitrate, lead and chloride) by 10 μg/100 mL nZVI-NPs was observed during reaction times of 12–24 hours, confirming the use of the NPs as a viable remediation strategy. The nZVI-NPs adsorbed 85.01% of phosphate, 99.51% of ammonia, 86.33% of nitrate, and 83.4% of chloride present in the samples initially, as well as removing 79.33% of the lead present within 24 hours.


 Please wait while we load your content...
Please wait while we load your content...