Porous carbon nanosheets functionalized with Fe3O4 nanoparticles for capacitive removal of heavy metal ions from water†
Abstract
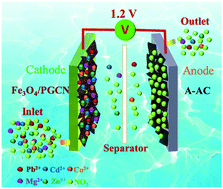
Development of cheap and high-efficiency electrode materials applied to capacitive deionization (CDI) technology is critically important for ensuring the safety of drinking water through capacitive removal of heavy metal ions. Here, we report ultrafine Fe3O4 nanoparticle (NP) functionalized porous graphitic carbon nanosheets (PGCNs) derived from sugarcane bagasse (Fe3O4/PGCNs) fabricated through a combined pyrolysis and incipient-wetness impregnation treatment approach. The as-proposed Fe3O4/PGCN has a large specific surface area of 1692.6 m2 g−1, a hierarchical porous structure and outstanding electroconductivity. Importantly, the functionalization of ultrafine Fe3O4 NPs (4–7 nm) on sugarcane bagasse derived PGCNs enables Fe3O4/PGCNs to have significantly enhanced electrosorption capacity, surface negative charge and wettability compared to PGCNs without modification, as a promising CDI electrode material for highly efficient decontamination of heavy metal ions from water. Here, Fe3O4/PGCNs as the cathode and amino-functionalized commercial activated carbon (A-AC) as the anode were used to construct a membrane-free hybrid capacitive deionization (HCDI) system displaying very high ion removal capacities (20.9 and 20.2 mg g−1), average removal rates (0.7 and 0.67 mg g−1 min−1) and removal efficiency (>95%) towards Pb2+ and Cd2+ with a concentration of 20 mg L−1 in water (pH = 5.5–6.5). The HCDI system made from Fe3O4/PGCN//A-AC also exhibits good regeneration performance, high recycling stability and strong ion removal capability towards a mixture of multiple heavy metal ions (Mg2+, Zn2+, Cu2+, Cd2+ and Pb2+) with a removal efficiency of 90–97%, indicating great application potential for drinking water purification.
- This article is part of the themed collection: Capacitive deionisation and electrosorption 2020


 Please wait while we load your content...
Please wait while we load your content...