Response of intestinal Gα subunits to nanopolystyrene in nematode Caenorhabditis elegans†
Abstract
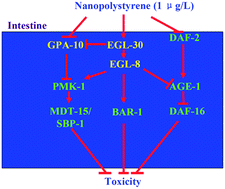
In organisms, Gα subunits transduce the signals from G protein-coupled receptors (GPCRs) to downstream cytoplasmic signaling pathways. However, the role of G proteins in regulating the response to nanoplastics remains largely unclear. Using Caenorhabditis elegans as an animal model, we identified intestinal Gα subunits involved in the control of response to nanopolystyrene (100 nm). After the exposure, nanopolystyrene in the range of μg L−1 could significantly increase the expression of egl-30 and decrease the expression of gpa-10. Intestine-specific RNAi knockdown of egl-30 induced susceptibility to nanopolystyrene toxicity, and intestine-specific RNAi knockdown of gpa-10 induced resistance to nanopolystyrene toxicity, suggesting that the alteration in intestinal EGL-30 and GPA-10 expressions mediated a protective response. Intestinal EGL-30 regulated the response to nanopolystyrene by activating the EGL-8/PLCβ. Moreover, in nanopolystyrene exposed nematodes, intestine-specific RNAi knockdown of egl-30 or egl-8 significantly decreased the expressions of pmk-1 encoding a p38 MAPK, bar-1 encoding a β-catenin transcriptional factor, and daf-16 encoding a FOXO transcriptional factor, and increased the expression of age-1 encoding a kinase acting downstream of insulin receptor DAF-2. Additionally, in nanopolystyrene exposed nematodes, intestine-specific RNAi knockdown of gpa-10 significantly increased the expressions of pmk-1 and its downstream signaling cascade of mdt-15-sbp-1. Therefore, intestinal Gα subunits (EGL-30 and GPA-10) were involved in the control of response to nanopolystyrene by regulating the downstream insulin, p38 MAPK, and/or Wnt signaling pathways. Our results highlight the crucial function of intestinal Gα subunits in response to nanoplastics in the range of μg L−1 in organisms.


 Please wait while we load your content...
Please wait while we load your content...