Insight into the size effect of Pd nanoparticles on the catalytic reduction of nitrite in water over Pd/C catalysts†
Abstract
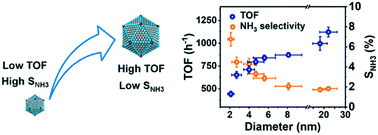
Catalytic denitrification over Pd-based catalysts under mild conditions has received significant attention as an emerging technology. However, the Pd particle size effect has been much less systematically investigated. Herein, carbon-supported Pd nanoparticles (NPs) with particle sizes ranging from 2.1 to 22.1 nm were synthesized and characterized using high-resolution transmission electron microscopy (HR-TEM), X-ray diffraction (XRD), CO-chemisorption, and X-ray photoelectron spectroscopy (XPS). Catalytic nitrite reduction experiments were conducted in a semi-batch reactor at room temperature. The results showed that the highest mass catalytic activity of 603.6 mg gPd−1 min−1 was obtained at the Pd size of 2.7 nm. The turnover frequency (TOF) and the N2 selectivity increased gradually with the increasing Pd NP size. Considering both the mass catalytic activity and N2 selectivity, the Pd catalyst with a size of approximately 5 nm had a better comprehensive performance. Mechanism studies showed that the enhanced TOF of larger Pd NPs could be ascribed to the formation of more β-hydride, which has a higher reducing activity than α-hydride. Compared with the terraces, the edges and corners of Pd NPs are the major active sites where ammonia is generated, most likely because of their low-coordination and electron-deficient properties. This indicated that the NO2− catalytic reduction is a size sensitive reaction in terms of both activity and selectivity, and may provide new insight into tailoring Pd NP size of Pd-based catalysts to improve catalytic activity and selectivity for nitrate and nitrite reduction in water.


 Please wait while we load your content...
Please wait while we load your content...