How does calcium drive the structural organization of iron–organic matter aggregates? A multiscale investigation†
Abstract
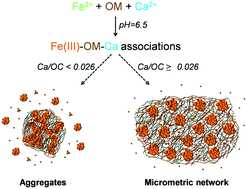
Iron–organic matter (Fe–OM) aggregates are a key factor in the control of pollutant mobility. Their physical and structural organization depends on the prevailing physicochemical conditions during their formation and on subsequent exposure to variations in porewater geochemistry. Among these conditions, calcium (Ca) could be a major parameter given its high concentrations in the environment and its affinity for OM. Mimetic environmental Fe–OM–Ca associations were synthesized at various Fe/organic carbon (OC) and Ca/Fe molar ratios using leonardite humic acid as the OM model. The impact of Ca on Fe–OM aggregates was studied by a combination of X-ray absorption spectroscopy, small angle X-ray and neutron scattering and imaging techniques (TEM, cryo-TEM and cryo-TXM). Iron phases are constituted of Fe(III)-oligomers, Fe(III)-nanoparticles and ferrihydrite (Fh), all bound to or embedded in OM. Iron phases exhibit a fractal organization with Fe-primary beads aggregated as Fe-primary aggregates (Fe-PA) which themselves are embedded in OM aggregates. For Ca/OC (mol mol−1) < 0.026, Fe-PA aggregate in a third level as an Fe-secondary aggregate. For Ca/OC ≥ 0.026, OM forms a large Ca-branched network in which Ca is bound as a dimer to OM carboxylic sites. Under such conditions, Fe-PA are distributed in the OM network, distant from each other. All these structural transitions are driven by Ca which partially screens the Fe–OM interactions. The formation of such a micrometric network should impact both the surface reactivity of the Fe phases and the mobility of Fe, OM and associated elements, notably in soil pores where they are produced under natural conditions.


 Please wait while we load your content...
Please wait while we load your content...