Criteria of active sites in nonradical persulfate activation process from integrated experimental and theoretical investigations: boron–nitrogen-co-doped nanocarbon-mediated peroxydisulfate activation as an example†
Abstract
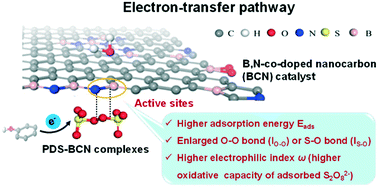
Carbon-catalyzed persulfate activation is a metal-free advanced oxidation process for abating aqueous organic micropollutants. Recently, the electron-transfer mechanism in the activation of peroxydisulfate (PDS) has attracted tremendous interest due to its unknown nonradical reaction pathways. The conventionally used atomic-scale descriptors of adsorption energy (Eads), O–O bond length (lO–O) and S–O bond length (lS–O) cannot accurately reflect the ability of the functionalities of PDS in its activation. In this work, a new descriptor, local electrophilicity index (ω), which represents the oxidative capacity of adsorbed S2O82−, was included to identify the intrinsic active sites in carbocatalysts via density functional theory calculations. To verify the reliability of the proposed criteria, the catalytic performances of a series of highly boronated and nitrogenated carbon nanotube/nanosheet composites (BCN-NT/NS) with tailored physicochemical properties were comparatively studied for activating PDS to degrade phenol. By integrating the computational and experimental results, the catalytic activity of BCN-NT/NS was determined to not only be related to the contents of heteroatom dopants (B and N), but also the positions of B and N in the co-doping configurations. This study offers reliable criteria for determining the intrinsic catalytic sites in carbocatalysts for the activation of PDS based on an electron-transfer mechanism, which assists the rational design of nanocarbons as advanced catalysts for metal-free oxidation and water remediation.
- This article is part of the themed collection: Environmental Science: Nano Cover Art


 Please wait while we load your content...
Please wait while we load your content...