Photocatalytic reduction of Cr(vi) by graphene oxide materials under sunlight or visible light: the effects of low-molecular-weight chemicals†
Abstract
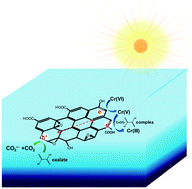
We present a new, rapid method to photocatalytically reduce the highly toxic Cr(VI) to Cr(III) by a graphene oxide (GO) photocatalyst utilizing renewable sunlight or visible light. We found that GO can photoreduce Cr(VI) (0.32 mM) with 35.8 ± 4.8% conversion in 120 min of solar irradiation and that oxalate significantly accelerates this reaction (85.6 ± 5.4% in 30 min). The photocatalytic reactivity of GO is comparable with or superior to that of P25 TiO2 under sunlight or visible light irradiation and is among the best in existing photocatalytic Cr(VI) reduction systems involving carbon-based nanomaterials. In contrast, the photoreduction of Cr(VI) using reduced GO greatly declines with 8.5 ± 1.4% conversion in 30 min. The efficacy of low-molecular-weight (LMW) chemicals follows the order: oxalate > EDTA > formate ≈ acetate ≈ methanol ≈ no addition. The measured molar ratio of oxalate consumed by reduced Cr(VI) is 1.6 ± 0.26, consistent with oxalate as an electron donor. The UV-visible absorbance spectrum indicates the formation of the oxalate–Cr(V) complex that could facilitate Cr(VI) photoreduction. Chromium speciation studies using the HPLC-ICP-MS method quantitatively confirmed that the increase of Cr(III) species was directly attributed to Cr(VI) photoreduction. The photocatalytic reduction by GO shows good stability in real wastewater samples and after three cycles of reaction. Our results indicate that GO is a simple and promising new photocatalyst toward Cr(VI) reduction.


 Please wait while we load your content...
Please wait while we load your content...