A 3D soil-like nanostructured fabric for the development of bacterial biofilms for agricultural and environmental uses
Abstract
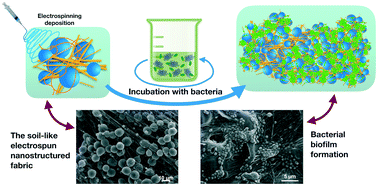
The present study aimed at creating a solid nanostructured scaffold suitable for the development of bacterial biofilms, and that enabled bacteria to successfully resist harsh nutritive and environmental conditions for potential applications. As a model system, we combined a self-standing electrospun nanostructured poly(ε-caprolactone)-based scaffold (EN-PCLS) with Burkholderia terricola cells. The scaffold structure was fabricated on purpose to include microbeads and nanofibres, and mimic the 3D morphological and spatial architecture of soil at the microscale and nanoscale, thus favouring the development of suitable biofilms. The resulting 3D framework displayed an extensive porosity and pore interconnectivity, and a honeycomb wax cell-like arrangement reproducing large cavities in soil. The bacteria first deposited a conditioning film onto the EN-PCLS to facilitate adhesion. The bacterial attachment was preferentially observed on the nanofibres with cells prevalently oriented along with them, and it persisted until late incubation under stirring. Notably, beads appeared bacteria-free if deprived of nanofibres on them. Over time, the interaction was improved in terms of stability upon the formation of appendages and release of extracellular polymeric substances (EPS). The more stable adhesion to the nanofibres enhanced the colonisation until the formation of flat bacterial aggregations in the nanofibrous areas inside the honeycomb wax cell-like frames of the EN-PCLS. Also, 3D micro- and macrocolonies were formed, and hung to nanofibres in between the microbeads of the honeycomb wax cell-like walls. Such microbial organisation evolved into a mature 3D biofilm within 7 d incubation, where bacteria formed densely packed layers of cells embedded into and coated with the EPS matrix that covered as a thick blanket the whole soil-like EN-PCLS. At the final stage of biofilm development (11 d), the biofilm and scaffold frameworks both appeared partially degraded, with the latter being probably utilised as a C-source. Hence, the observed soil-like 3D nanostructured architecture proposed here has successfully enabled soil bacteria to develop biofilms on more natural supports than in traditional studies. Thus, this strategy seems promising in creating scaffolds to be used as successful nanobiotechnological carriers for applications in both agriculture (nutrient supply and control of plant diseases) and environment (bioremediation of polluted soils and wastewater).
- This article is part of the themed collection: Environmental Science: Nano Cover Art


 Please wait while we load your content...
Please wait while we load your content...