The surface hydroxyl and oxygen vacancy dependent Cr(vi) adsorption performance of BiOCl†
Abstract
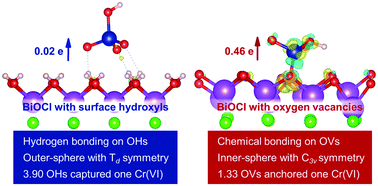
The migration and bioavailability of Cr(VI) are determined by its adsorption behavior, which is sensitive to the coordination environment and to the physicochemical properties of mineral surfaces, especially natural surface defects, such as surface hydroxyls (OHs) and oxygen vacancies (OVs). However, natural minerals with high percentages of surface OHs and OVs are not available, concealing the quantitative relationship between surface OHs and OVs and Cr(VI) adsorption on minerals. With regard to the easy tuning of OHs and OVs on the 001 surface of BiOCl (BiOCl-001), herein we systematically check how surface OHs and OVs influence Cr(VI) adsorption on BiOCl-001, using density functional theory calculations, in situ attenuated total reflectance Fourier transform infrared spectroscopy, and synchrotron-based Cr K-edge extended X-ray absorption fine structure spectroscopy, aiming to elucidate the contribution of surface OHs and OVs to Cr(VI) adsorption on minerals at a molecular level. The results revealed that 3.90 OHs captured one Cr(VI) in a hydrogen bonded adsorption and outer-sphere model with subtly distorted Td symmetry, and 1.33 OVs anchored one Cr(VI) by forming a Cr–O–Bi bond to produce a monodentate mononuclear inner-sphere surface complex with C3v symmetry, along with the partial reduction of Cr(VI) by unpaired electrons located in the OVs on BiOCl-001. This difference would intensely affect the mobility and long-term fate of Cr(VI) species. This study clarifies the surface defect dependent adsorption behavior of Cr(VI), and sheds light on the environmental effects of surface OHs and OVs of minerals.


 Please wait while we load your content...
Please wait while we load your content...