Nanodenitrification with bimetallic nanoparticles confined in N-doped mesoporous carbon†
Abstract
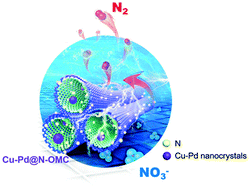
Nitrate and phosphorous contamination owing to increasing fossil fuel consumption and the use of modern fertilizers is the leading cause of the eutrophication and deterioration of water quality worldwide. Herein, we introduced an N-doped ordered mesoporous carbon-based skeleton with Cu–Pd nanocrystals (Cu–Pd@N-OMC) for the electrocatalytic reduction of NO3− to dinitrogen (N2). Laboratory experiments demonstrated high NO3− removal (91%) and N2 selectivity (97%) for eutrophic water treatment. The nanocatalyst also remained highly active over 15 experimental cycles. Metal–organic frameworks (MOFs) induced dispersion and the embedment of small Cu–Pd nanocrystals (4–9 nm) in the carbon mesochannels. Moreover, the metal loading was as low as 5 wt% with the Cu/Pd ratio at 4/1. The results suggested that the electrocatalytic reactivity could be fine-tuned by the size and crystallinity of the Cu–Pd nanoparticles through controlling the pyrolysis temperature. The high-density Cu–Pd nanocrystals promoted rapid hydrogen adsorption to cleave the N–O bond for high N2 selectivity. The incorporation of MOFs and OMC improved the dispersion and stabilization of metallic nanoparticles for prolonged electrocatalytic activity and durability.


 Please wait while we load your content...
Please wait while we load your content...