Study of the toxicity of ZnO nanoparticles to Chlorella sorokiniana under the influence of phosphate: spectroscopic quantification, photosynthetic efficiency and gene expression analysis†
Abstract
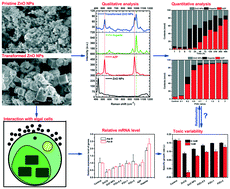
Zinc oxide nanoparticles (ZnO NPs) are one of the most abundantly applied nanomaterials in nanotechnology-based industries, and recent research continues to highlight their potential eco-toxicity especially to aquatic environments. When they enter into a water environment, their physicochemical transformations may result in largely unknown end products and induce additional potential toxicity. Although previous studies have documented the transformation of ZnO NPs, there is still a lack of the in-depth understanding of the corresponding variant toxicities of the pristine and transformed ZnO NPs to aquatic organisms, particularly demanding the quantitative analysis of the physicochemical transformations to distinguish their contributions in the toxicity assessment. For this purpose, therefore, we initiated a study of the toxicity of transformed ZnO NPs in phosphate containing water to a model aquatic microalga, i.e., Chlorella sorokiniana, with the aid of spectroscopic tools for characterization and quantification of the physicochemical transformations, and we scrutinized the toxicity variations of ZnO NPs transformed in different P/Zn molar ratios. The possible toxicity mechanisms were investigated, especially those related to the influence of photosynthesis efficiency of the algae cells. As a result, we found that the toxicity of transformed ZnO NPs in phosphate water to the green algae was lower than that of the pristine ZnO NPs. With spectroscopic tools such as XRD and Raman spectroscopy, we made both qualitative and quantitative assessments of the physicochemical changes of the ZnO NPs and revealed that for the pristine ZnO NP group, the toxicity stemmed mainly from the release of zinc ions from the pristine ZnO NPs, while in the presence of phosphate, the neoformation of the nanoparticles played a critical role, leading to the overall reduced toxicity due to the less toxic amorphous zinc phosphate (AZP) and hopeite (Zn3(PO4)2·4H2O) in the transformed compounds. Besides, the mechanism of the toxicity of phosphate induced transformations of ZnO NPs to C. sorokiniana was also verified by the analysis of gene expression involved in photosynthesis. Our results showed that the expressions of the photosynthesis associated genes were depressed by the treatment of pristine ZnO NPs, while the expressions of the related genes were up-regulated in the groups of hopeite and AZP, indicating that the soluble phosphate had profound effects on algal photosynthesis, and thus affected the algal toxicity of ZnO NPs in the aqueous environment. These newly acquired results will certainly elaborate our knowledge on the fate and effects of ZnO NP transformation in aquatic environments.


 Please wait while we load your content...
Please wait while we load your content...