Single-molecule determination of the phase- and facet-dependent adsorption of alginate on iron oxides†
Abstract
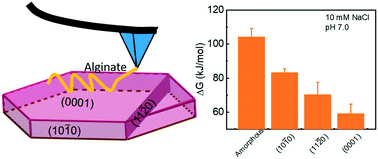
The formation of iron oxide mineral–organic associations can regulate long-term preservation of soil organic matter (SOM) by providing shelter for SOM from degradation through adsorption on different crystalline/amorphous phase surfaces. However, little is known about the energetic basis for the phase- and facet-dependent SOM–iron oxide interactions at the molecular scale. Here, we use a representative organic molecule, alginate, which is present ubiquitously in soils, and investigate its adsorption on the surfaces of amorphous iron oxides (FexOy) or hematite (α-Fe2O3) by calculating the Kelvin potential difference and measuring the binding energy (ΔGB) between an alginate molecule and a specific surface of the hematite or amorphous FexOy phase by atomic force microscopy (AFM). Single-molecule determination of alginate–amorphous FexOy/hematite interactions provides mechanisms of the origin of the phase- or facet-dependent adsorption by systematically changing the pH and ionic strength/compositions of environmentally relevant reaction solutions. We show that the adsorption capacities of alginate follow the order amorphous FexOy > the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) > (11
0) > (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) > (0001) faces of hematite. These molecular-scale investigations may improve our understanding of the phase-dependent or face-specific adsorption of SOM on other soil minerals in aqueous environments and potentially predict the preservation mechanisms and the fate of SOM.
0) > (0001) faces of hematite. These molecular-scale investigations may improve our understanding of the phase-dependent or face-specific adsorption of SOM on other soil minerals in aqueous environments and potentially predict the preservation mechanisms and the fate of SOM.
- This article is part of the themed collection: Nano-bio interactions


 Please wait while we load your content...
Please wait while we load your content...