Incorporation of Pb(ii) into hematite during ferrihydrite transformation†
Abstract
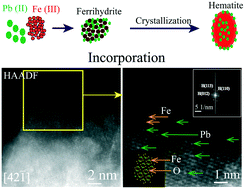
Ferrihydrite is ubiquitous in natural environments, and its transformation significantly influences the fate of heavy metals. Although Pb(II) adsorption on iron oxides has been extensively studied, there is still a knowledge gap on the fate of Pb during the dynamic processes of iron oxide transformation. In this study, a set of wet chemistry experiments, spherical aberration corrected scanning transmission electron microscopy (Cs-STEM) integrated with energy dispersive X-ray spectroscopy (EDS), X-ray diffraction (XRD), and X-ray absorption spectroscopy (XAS) were used to unravel Pb interactions with iron oxides during ferrihydrite transformation processes under abiotic conditions. Wet chemistry experiments, STEM-EDS elemental mapping and line scans, and quantitative analysis suggested that Pb penetrated into hematite nanoparticles during the transformation processes. STEM analysis at sub-nanoscales, XRD fine scans, and XAS analysis provided evidence of Pb incorporation into the crystal structures of hematite nanoparticles, in which Pb distributed along the zone axis and enlarged the lattice distance of hematite. These results advanced our understanding of the dynamic interactions of Pb with iron oxides by offering new perspectives about the critical roles of chemical speciation, nano-scale spatial distribution, and atomic coordination environments in controlling the geochemical dynamics of heavy metals.
- This article is part of the themed collections: Environmental Remediation and Environmental fate of nanomaterials


 Please wait while we load your content...
Please wait while we load your content...