Transformation and uptake of silver nanoparticles and silver ions in rice plant (Oryza sativa L.): the effect of iron plaque and dissolved iron†
Abstract
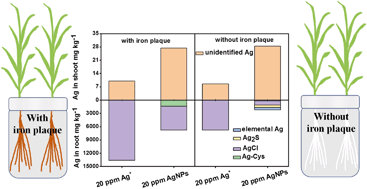
The wide use of silver ions (Ag+) and silver nanoparticles (AgNPs) and their inevitable release into the environment have caused great concern over their environmental and health risks. The understanding of their uptake and accumulation by plants, especially food crops, is important to evaluate their risks. We reported herein the distinct accumulation and translocation characteristics of Ag+ and AgNPs in a rice plant (Oryza sativa L.) at exposure doses of 2, 5, 10, and 20 mg L−1, as well as the critical impact of iron plaque on their assimilation by the plant. As revealed by silver K-edge X-ray absorption near-edge structure (XANES), Ag+ in/on the roots was exclusively transformed into AgCl with Cl− in hydroponic medium. As a result, AgCl maintained Ag+ on the root surface, lowering its bioavailability and translocation (with translocation factors, TFs, from 0.44 × 10−3 to 1.79 × 10−3). On the contrary, AgNPs were assimilated and translocated more effectively (with TFs from 2.44 × 10−3 to 1.23 × 10−2) in comparison to Ag+, although a fraction was transformed into ionic silver species. XANES demonstrated that the iron plaque present on the rice roots could oxidize AgNPs into ionic Ag species, such as AgCl and Ag+–thiol complexes, and increase the uptake and translocation of silver in the rice plant. Compared to iron plaque, dissolved iron was less effective in oxidizing AgNPs. Our findings highlighted the importance of Ag species interconversion and iron plaque in the uptake and translocation of Ag, providing useful data for an improved risk assessment of AgNPs and Ag+ under environmental scenarios.


 Please wait while we load your content...
Please wait while we load your content...