Immobilization of mercury by nano-elemental selenium and the underlying mechanisms in hydroponic-cultured garlic plant†
Abstract
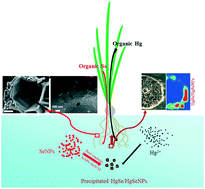
In mining areas, nano-sized elemental selenium (SeNPs), naturally produced through reduction of SeO32− by abiotic and biotic processes, usually co-exists with Hg in the environment. Little is known about the fate of SeNPs and how they affect the bioavailability and toxicity of Hg towards plants. In this study, the fate of SeNPs and Hg, and the effect of SeNPs on the mobilization of Hg in a hydroponic-cultured plant were investigated. It was found that SeNPs were less phytotoxic and had stronger capacity for Hg sequestration than SeO32−and SeO42−. Synchrotron radiation X-ray fluorescence (SR-XRF) and X-ray absorption near edge structure (XANES) spectroscopy revealed that SeNPs largely captured Hg2+via forming HgSe and HgSeNPs to prevent Hg2+ from entering the root stele, thus inhibiting translocation and accumulation of Hg in the aerial parts. Apart from immobilizing Hg, SeNPs also promoted the conversion of Hg2+ to less toxic RS–Hg–SR and R–Hg–R binding forms in plants. Meanwhile, a portion of SeNPs was transformed into SeMet stored as the nutrient Se source in plant tissues. This study provides insights into the reaction and fate of SeNPs and Hg in water-plant systems, which implies the significance of SeNPs in modulating the fate and toxicity of Hg in mining areas.


 Please wait while we load your content...
Please wait while we load your content...