Sulfide and ferrous iron preferentially target specific surface O-functional groups of graphene oxide: implications for accumulation of contaminants†
Abstract
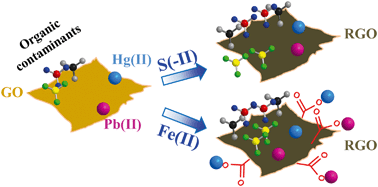
Carbonaceous nanomaterials, such as graphene oxide (GO), are susceptible to environmental reduction that affects their fate and impact in aquatic environments. Here, we demonstrate that sulfide and ferrous iron, the most prevalent natural reductants, significantly alter the surface properties of GO, which can enhance the accumulation of both organic and inorganic contaminants on GO. Strikingly, these two reductants target different surface O-functional groups while diminishing the overall surface O-functionality of GO (i.e., C/O ratio) to a similar extent at a given reductant concentration. Hence, the two reductants have distinctly different effects on the adsorption of organics vs. heavy metals onto GO. At the same reductant concentration, iron and sulfide reduction increases the adsorption of naphthalene, 1,2,4,5-tetrachlorobenzene and 2,4-dichlorophenol comparably, as the adsorption of polar and nonpolar organic contaminants on GO is driven primarily by hydrophobic effects and π–π interactions, which correlate reasonably with the C/O ratio of GO. Interestingly, while iron reduction significantly promotes the adsorption of heavy metals, sulfide reduction has essentially no effect. Iron reduction apparently increases the abundance of GO carboxyl groups and consequently promotes the binding of mercury and lead via inner-sphere complexation. The increasing content of carbonyl groups induced by sulfide reduction may enhance metal adsorption on GO, but this effect is probably offset by the simultaneous decrease of carboxyl groups and decreasing negative charges of GO. This research highlights the importance of understanding the specific reduction pathway of carbonaceous nanomaterials when assessing their impact on the environmental behavior of metals.


 Please wait while we load your content...
Please wait while we load your content...