Validation of a field deployable reactor for in situ formation of NOM-engineered nanoparticle corona†
Abstract
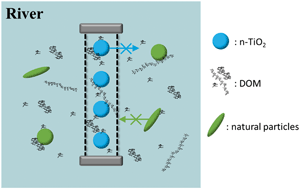
Despite the numerous studies about the sorption of dissolved organic matter (DOM) onto nanoparticles, the extrapolation of laboratory results to environmental conditions is currently impossible. Indeed, the complex dynamics of DOM under variable environmental conditions are not completely reproducible under control conditions. In this study, we propose a different approach by exploring a method for exposing nanoparticles to realistic environmental conditions in natural river water by using dialysis membranes as passive reactors. Inside this reactor, the complexity and the temporal variability of a large number of environmental parameters (DOM structure and composition, temperature, inorganic ions, pH, etc.) are reproduced, while colloidal and particulate interferences remain separated. To verify this assumption, we determined the concentration of the water components and nanoparticles (n-TiO2, 20–50 nm) inside and outside the reactor before and after exposure to river water. In river water, more than 90% of the n-TiO2 nanoparticles remained inside the reactor while DOM retained its molecular composition/characteristics after passing through the membrane (DOC, fluorescence EEM, and FT-ICR MS). For most elements and anions, the concentrations inside and outside the reactor did not differ, indicating a good permeability for inorganic constituents (IC, ICP-OES); however, the concentrations of Al, Fe, Mn, and nitrate were lower. Membrane fouling, in terms of pore size distribution, was investigated using NMR relaxometry and AFM in fluid mode; no significant reduction in pore size was observed under the applied conditions during seven days of exposure. Finally, ATR-FTIR and CHNS analysis of n-TiO2 before and after exposure to the river water revealed that sorption of DOM occurred under field conditions. Therefore, we could demonstrate the validity and the potential of this method.


 Please wait while we load your content...
Please wait while we load your content...