Factors determining the toxicity of engineered nanomaterials to Tetrahymena thermophila in freshwater: the critical role of organic matter†
Abstract
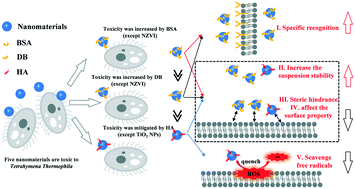
The environmental behavior and toxicity of nanomaterials can be significantly affected by the ubiquitous natural organic matter (NOM) of aquatic environments. However, general mechanisms governing the effects of different organic matter samples on nanomaterials remain elusive at present. Thus, the environmental behavior and toxicity of organic matter with different components or conformation, such as humic acid (HA), bovine serum albumin (BSA) and denatured BSA (DB), on five nanomaterials (i.e., Ag nanoparticles (NPs), polystyrene nanoparticles (PS NPs), TiO2 NPs, nano-zerovalent iron (NZVI), and multiwall carbon nanotubes (MWCNTs)) to Tetrahymena thermophila (T. thermophila) were studied. BSA and DB were found to significantly increase (up to 99%) the bioaccumulation of nanomaterials (Ag NPs and PS NPs) in T. thermophila by specific recognition. This increase was lower in DB-coated nanomaterials, compared to BSA-coated nanomaterials, revealing the importance of protein conformation in the specific recognition process. The toxicity results revealed that different organic matter samples mostly affected Ag NPs and NZVI (toxicity by dissolved metal ions) by altering their biological uptake. In the case of nanomaterials whose toxicity was dominated by suspended particles, the organic matter could alter the toxicity of these materials by changing their surface properties and their state in environmental media, and by scavenging free radicals, instead of altering the bioaccumulation process. In general, the toxicity of HA-coated nanomaterials was significantly lower than those of DB- or BSA-coated nanomaterials. Our results suggest that it is essential to consider the type and characteristics of the organic matter when evaluating the toxicity of NPs, since this represents a more realistic state of exposure.


 Please wait while we load your content...
Please wait while we load your content...