Sorption–desorption of dimethoate in urban soils and potential environmental impacts†
Abstract
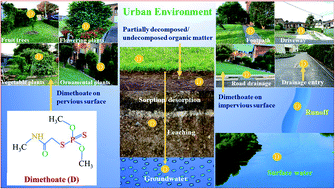
The environmental fate and impact of dimethoate application in the urban environment were assessed in nine selected soils. The pseudo-second-order kinetics model described the kinetics of dimethoate sorption very well in the urban soils exhibiting two distinct phases, an initial partitioning into clay surfaces and soil organic matter, and eventual diffusion into soil micropores. Dimethoate sorption in the urban soils followed the Freundlich model with an R2 value of 0.94–0.99, suggesting a multi-layered sorption on the heterogeneous surfaces. Sorption of dimethoate in the soils was influenced by clay, silt, organic matter, carboxyl and alkyl groups, and Al and Fe oxides. The undecomposed or incompletely decomposed organic matter present in the soils greatly reduced the sorption and enhanced desorption. The calculated lower values for Freundlich constant (KF) indicate the high mobility of dimethoate in the selected soils. Also, the values of groundwater ubiquity score (GUS), leachability index (LIX), hysteresis index (HI), and coefficient of distribution (Kd) for dimethoate in the soils clearly suggest that the insecticide is prone to leaching out significantly from the soil surface to groundwater. Moreover, the surface runoff from impervious places in the urban environment can be considered as a direct source of groundwater contamination, thereby affecting the quality of potable water besides posing a threat to non-target organisms of ecological importance and food safety. Thus, the present novel study suggests that the application of dimethoate in the urban environment having impervious surfaces must be judicious in order to minimize the potential human and ecological health risks.


 Please wait while we load your content...
Please wait while we load your content...