Interactions of emerging contaminants with model colloidal microplastics, C60 fullerene, and natural organic matter – effect of surface functional group and adsorbate properties†
Abstract
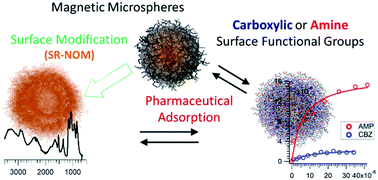
Surface adsorption of two commonly detected emerging contaminants, amlodipine (AMP) and carbamazepine (CBZ), onto model colloidal microplastics, natural organic matter (NOM), and fullerene nanomaterials have been investigated. It is found that AMP accumulation at these colloidal–aqueous interfaces is markedly higher than that of CBZ. Measurements of surface excess and particle zeta potential, along with pH-dependent adsorption studies, reveal a distinct influence of colloidal functional group on the adsorption properties of these pharmaceuticals. AMP shows a clear preference for a surface containing carboxylic group compared to an amine modified surface. CBZ, in contrast, exhibit a pH-dependent surface proclivity for both of these microparticles. The type of interactions and molecular differences with respect to structural rigidity and charge properties explain these observed behaviors. In this work, we also demonstrate a facile approach in fabricating uniform microspheres coated with NOM and C60 nanoclusters. Subsequent binding studies on these surfaces show considerable adsorption on the NOM surface but a minimal uptake of CBZ by C60. Adsorption induced colloidal aggregation was not observed. These findings map out the extent of contaminant removal by colloids of different surface properties available in the aquatic environment. The methodology developed for the adsorption study also opens up the possibility for further investigations into colloidal–contaminant interactions.
- This article is part of the themed collection: Urgent communications in RSC Environmental Science journals


 Please wait while we load your content...
Please wait while we load your content...