Reactions of chlorinated ethenes with surface-sulfidated iron materials: reactivity enhancement and inhibition effects†
Abstract
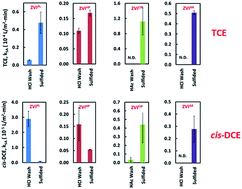
Recent studies on the use of controlled sulfur amendment to improve the reactivity and selectivity of zerovalent iron (ZVI) in reductive dechlorination reactions have generated renewed interest in ZVI-based remediation materials. However, existing studies have focused on the reactions between trichloroethene (TCE) and lab-synthesized ZVI, and the applicability of sulfidation to ZVIs with different material characteristics for reductive dechlorination of chloroethenes such as tetrachloroethene (PCE) and cis-dichloroethene (cis-DCE) has not been systematically examined. In this study, four ZVI materials from commercial sources having different sizes and morphological and compositional characteristics were subjected to various sulfidation treatments and were assessed in batch reactions with PCE, TCE, or cis-DCE. Sulfur amendment induces modest increases in PCE degradation rates and steers reactions towards a cleaner pathway that has minimum accumulation of partially dechlorinated intermediates. In the case of cis-DCE, bifurcating outcomes were observed that include enhancement effects for two high-purity ZVIs and inhibitory effects for two ZVIs possessing low levels of metal impurities. Further investigations based on controlled metal dosing reveal that the trace metals commonly present in cast iron or recycled metal scraps, such as Cu and Ni, can act as adventitious catalysts for cis-DCE reduction. Sulfidation results in poisoning of these catalytic ingredients and accounts for the adverse effect observed with a subset of ZVIs. Collectively, this study confirms enhanced degradation of highly chlorinated ethenes (PCE and TCE) by sulfidation of ZVIs from diverse origins; nonetheless, the effects of sulfidation can be highly variable for the less chlorinated ethenes due to differences in the material characteristics of ZVI and the predominant dechlorination pathways.
- This article is part of the themed collection: Halogenated (semi)volatile organic compounds (“X(S)VOCs”)


 Please wait while we load your content...
Please wait while we load your content...