Compound-specific chlorine isotope fractionation in biodegradation of atrazine†
Abstract
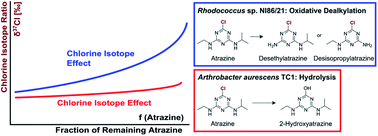
Atrazine is a frequently detected groundwater contaminant. It can be microbially degraded by oxidative dealkylation or by hydrolytic dechlorination. Compound-specific isotope analysis is a powerful tool to assess its transformation. In previous work, carbon and nitrogen isotope effects were found to reflect these different transformation pathways. However, chlorine isotope fractionation could be a particularly sensitive indicator of natural transformation since chlorine isotope effects are fully represented in the molecular average while carbon and nitrogen isotope effects are diluted by non-reacting atoms. Therefore, this study explored chlorine isotope effects during atrazine hydrolysis with Arthrobacter aurescens TC1 and oxidative dealkylation with Rhodococcus sp. NI86/21. Dual element isotope slopes of chlorine vs. carbon isotope fractionation (ΛArthroCl/C = 1.7 ± 0.9 vs. ΛRhodoCl/C = 0.6 ± 0.1) and chlorine vs. nitrogen isotope fractionation (ΛArthroCl/N = −1.2 ± 0.7 vs. ΛRhodoCl/N = 0.4 ± 0.2) provided reliable indicators of different pathways. Observed chlorine isotope effects in oxidative dealkylation (εCl = −4.3 ± 1.8‰) were surprisingly large, whereas in hydrolysis (εCl = −1.4 ± 0.6‰) they were small, indicating that C–Cl bond cleavage was not the rate-determining step. This demonstrates the importance of constraining expected isotope effects of new elements before using the approach in the field. Overall, the triple element isotope information brought forward here enables a more reliable identification of atrazine sources and degradation pathways.
- This article is part of the themed collection: Halogenated (semi)volatile organic compounds (“X(S)VOCs”)


 Please wait while we load your content...
Please wait while we load your content...