Predicting Cr(vi) adsorption on soils: the role of the competition of soil organic matter†
Abstract
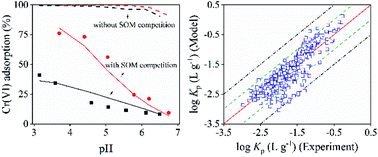
Cr(VI) has posed a serious risk for the environment and human beings because of its pollution and toxicity. It is essential to understand the equilibrium behavior of Cr(VI) in soils. In this study, the adsorption of Cr(VI) on fourteen soils was studied with batch experiments and quantitative modeling. The batch experiments included the adsorption edge and adsorption isotherm experiments, investigating the adsorption of Cr(VI) with varying soil properties, solution pH, and initial Cr(VI) concentrations. The experimental data were then modeled using the surface complexation models in Visual MINTEQ of CD-MUSIC by considering the adsorption of Cr(VI) and ions onto Fe (hydr)oxides and Al (hydr)oxides, and the Stockholm Humic Model and the fixed charge site model by accounting for the adsorption of the cations to soil organic matter and clay, respectively. Particularly, the modeling method of this study introduced an important parameter RO− to account for the amount of soil organic matter irreversibly adsorbed on soil minerals. Overall, the model predicted reasonably well for the equilibrium partition of Cr(VI) under various conditions with a root-mean-square-error of 0.35 for the adsorption edge data and 0.19 for the adsorption isotherm data. According to the model calculations, ferrihydrite dominated the binding of Cr(VI) at pH of 3.0–7.0. The content of ferrihydrite and reactive soil organic matter was found to be the main factor influencing RO−. The modeling results help to understand and predict Cr(VI) adsorption on different soils and are beneficial to environmental risk assessment and pollution remediation.
- This article is part of the themed collection: Environmental Science: Processes & Impacts Cover Art


 Please wait while we load your content...
Please wait while we load your content...