Methyl bromide production from dissolved organic matter under simulated sunlight irradiation and the important effect of ferric ions†
Abstract
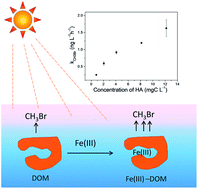
The oceans play an important role in the biogeochemical cycle of methyl bromide (CH3Br), as not only the sinks but also the sources. However, many uncertainties exist regarding the way of CH3Br generation in the marine environment. To illustrate the possibility of photochemical formation of CH3Br in saline water, its generation in bromide aqueous solution containing humic acid (HA) and ferric ions (Fe(III)) was investigated. CH3Br was obviously generated after irradiation, and its amounts increased with increasing HA concentration from 0.82 to 12.2 mgC L−1. Fe(III) significantly promoted CH3Br production, and the described production process in the presence of Fe(III) was pH-dependent, decreasing with the increase of pH. Finally, the concentrations of CH3Br in natural coastal seawater and seawater with HA were measured, and the results showed that CH3Br was significantly generated under irradiation. Our results suggest that the photochemical process of dissolved organic matter may be one source of CH3Br in the marine environment.
- This article is part of the themed collection: Halogenated (semi)volatile organic compounds (“X(S)VOCs”)


 Please wait while we load your content...
Please wait while we load your content...