Effect of the irrigation water type and other environmental parameters on CeO2 nanopesticide–clay colloid interactions†
Abstract
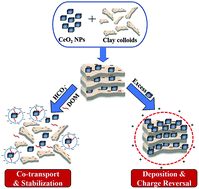
In this work, the stability and aggregation behaviour of CeO2 nanoparticles (NPs) was investigated to predict their fate in the agricultural environment. For this, the aggregation kinetics of CeO2 NPs was studied under varying pH, ionic strength (IS), dissolved organic matter (DOM) and carbonate concentrations in the presence of clay. Furthermore, different types of irrigation water have been used to check the fate of CeO2 nanoparticles (NPs) in complex aqueous matrices. The results show that critical coagulation concentration (CCC) values obtained for CeO2 NPs, i.e. 26.5 mM and 7.9 mM for NaCl and CaCl2 respectively, drastically decreased to 16.2 mM and 1.87 mM in the presence of bentonite clay colloids, which may lead to their deposition within the soil matrix. However, the presence of bicarbonate ions (0.1–2 mM) along with DOM (1–20 mg L−1) may result in their stabilization and co-transport of CeO2 NPs with clay in water bodies having low ionic strength. It was also observed that the negative charge of a bentonite clay suspension was completely reversed with an increase in CeO2 concentration by 37.5 times. The critical charge reversal concentration value was 284.4 mg L−1 in Milli-Q water whereas values were observed to be 680 mg L−1 in synthetic-soft water, followed by natural river water (867 mg L−1) and synthetic-hard water (910 mg L−1). The synergistic effect of temperature and ionic strength was observed on the aggregation behaviour of CeO2 NPs in environmental water samples of varying composition.
- This article is part of the themed collection: Environmental Science: Processes & Impacts Cover Art


 Please wait while we load your content...
Please wait while we load your content...