Ultrafast kinetics in a phase separating electrode material by forming an intermediate phase without reducing the particle size†
Abstract
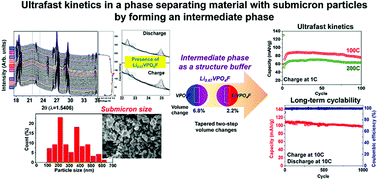
A fast charging/discharging capability in electrode materials is one of the essential properties of Li-ion batteries. A well-known approach for improving the kinetics of electrode materials is minimization of the particle size to the nanoscale, especially for phase separating materials such as LiFePO4 and Li4Ti5O12. Here, we report on ultrafast kinetics in a phase separating material with submicron particles enabled by the formation of an intermediate phase during discharge even without minimizing the particle size. The intermediate phase during (dis)charging acts as a structural buffer between the Li-rich and Li-poor phases, leading to reduced mechanical stress/strain on the interface in a particle, which can suppress structural defects, and a substantial reduction in the phase transformation barrier, which can make the electrochemical reaction of particles in the electrode homogenous. As a result, the resulting phase separating material can deliver ∼70 mA h g−1 at a 200C discharge rate (18 s for full discharge) with a 1C charge rate, and prolonged cycling stability for 1000 cycles even though it has a wide particle size distribution from 50 nm to 600 nm with an average of ∼300 nm. This finding will provide a new avenue for achieving fast electrochemical kinetics with a high volumetric energy density in electrode materials for high performance Li-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...