All ceramic cathode composite design and manufacturing towards low interfacial resistance for garnet-based solid-state lithium batteries†
Abstract
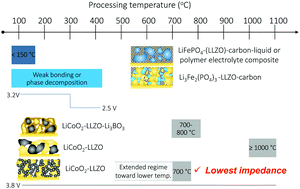
The critical factors that determine the performance and lifetime of solid-state batteries (SSBs) are driven by the electrode–electrolyte interfaces. The main challenge in fabricating all-oxide cathode composites for garnet-based SSBs has been lowering the thermal processing window in which both good contact and low interfacial resistance can be achieved. Here, we report an alternative ceramic processing strategy that enables the fabrication of all-oxide composite cathodes at an unusually low processing temperature without the use of extra sintering additives or a fluid electrolyte (polymer-gel or liquid electrolyte). We present specific examples of the most common LiFePO4 and LiCoO2 cathodes with a Li-garnet (Li7La3Zr2O12, LLZO) solid-electrolyte. We demonstrate an infiltration step to directly synthesize the LiCoO2 cathode from metal salts in a porous LLZO scaffold, resulting in the formation of a composite cathode such as LiCoO2–LLZO on top of a dense LLZO solid electrolyte at a low processing temperature of 700 °C. A promising discharge capacity of 118 mA h g−1 (3–4.05 V) with a low interfacial resistance of 62 Ohm cm2 is realized for LiCoO2 with a lithium anode, whereas critical phase instabilities for LiFePO4 are uncovered. Our findings encourage a move away from synthesis techniques that employ particle mixing and sintering to fabricate composites. We provide a blueprint for circumventing adverse interphase reactions according to chemistry and ceramic thermal processing budgets in the preparation of these ceramic interfaces as well as for increasing the number of reaction sites for high-performing composite cathodes for Li-garnet SSBs. In addition, the ceramic methods presented are scalable and mass manufacturable for the large-scale production of such composite cathodes for future industry.
- This article is part of the themed collection: Battery science and technology – powered by chemistry


 Please wait while we load your content...
Please wait while we load your content...